NEW BSC2-2 Dual Channel Interface Slice Chamber
This new interface mode brain slice chamber is a dual version of the BSC2 designed to maintain isolated, living tissues in vitro in ‘interface’ mode and allow stable electrophysiological recordings to be made from the preparation. The temperature is controlled by a low noise proportional control heating unit, the PTC03 or in cases where only incubation or non-ephys pharmacology is required, with a basic heating unit, the PTC04.
Features:
- ‘Interface’ method of slice maintenance.
- Two completely independent channels.
- Use entire surface for multiple slices or reduced area with templates.
- Single feed channel for perfusion solution.
- Proportional temperature controller (PTC03) with low noise performance used for heating.
- Optional basic temperature controller (PTC04) for interface slice incubation or pharmacology only.
The chamber lower section is similar to the BSC2 for temperature control and oxygenation. Pre-oxygenated medium enters the main body of the chamber through a feed line, one to each well. This fine bore PTFE tube which spirals in the heated distilled water in the lower part of the chamber enters the upper part of the chamber. Here any bubbles in the line break and allow perfusion fluid to flow smoothly towards the slices.
[su_lightbox type=”image” src=”/wp-content/uploads/2017/02/14534_BSC2-2-645×516-1.jpg” mobile=”yes” class=””] [/su_lightbox]
[/su_lightbox]
Acrylic templates allow the option for use of a single well or can be custom designed. The templates are positioned on the chamber acrylic base and optionally silicone grease is used to make the seal on the bottom. Typical dead space volume for the single well is approx. 200µl. The length of PTFE tubing carrying the perfusion solution to the wells has a volume of approx. 400µl. The flow rate of the perfusion fluid is adjusted at the exit well by means of lens tissue which is used to wick solution out of the exit port. By adjusting the over-hang the flow rate is effectively adjusted and determines the characteristics of the meniscus of fluid around the slice preparation.
As with all interface preparations, the high oxygen tension is maintained by bubbling a 95% oxygen, 5% carbon dioxide gas mixture through a ceramic bubbler located in the lower heated part of the chamber. This moistened and warmed gas mixture enters the upper part of the chamber via port-holes and is then deflected by an acrylic cover across to the centrally located slice preparation. The temperature in the upper chamber is maintained by ensuring that the medium and moistened gas mixture enter at the required temperature. This is dependent on the temperature of the lower chamber body which is warmed by a heating element controlled by the Proportional Temperature Controller, PTC03. An optional monitor sensor allows the upper chamber temperature to be checked when required. A more basic version of the controller PTC04 can be used instead where noise control is not an issue as with pre-incubation of slices in interface mode or for pharmacology experiments.
[su_lightbox type=”image” src=”/wp-content/uploads/2017/02/14535_BSC2-2_Top_View-645×489-1.jpg” mobile=”yes” class=””]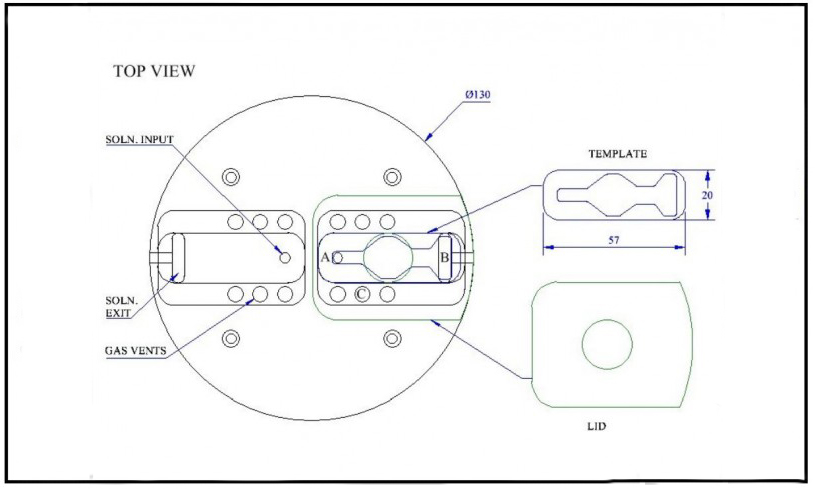 [/su_lightbox]
[/su_lightbox]
BSC2-2 and template system The channel within the template is diamond shaped for optimum flow and low dead space volume. The blue line opposite is a PTFE tube which spirals in the lower chamber before delivering the heated perfusate to the upper chamber, shown for one of the two independent wells of the BSC2-2. Ceramic aerator delivers fine bubbles of carbogen mixture into warmed distilled water in base of chamber. The warmed, humidified gas passes through port holes into the upper chamber and is then deflected by a lid and concentrated above the interface slice located in the template. Proportional temperature controller is used to maintain selected temperature via sensors and heating element located in lower chamber.
[su_lightbox type=”image” src=”/wp-content/uploads/2017/02/14536_BSC2-2_Side_View-645×485-1.jpg” mobile=”yes” class=””]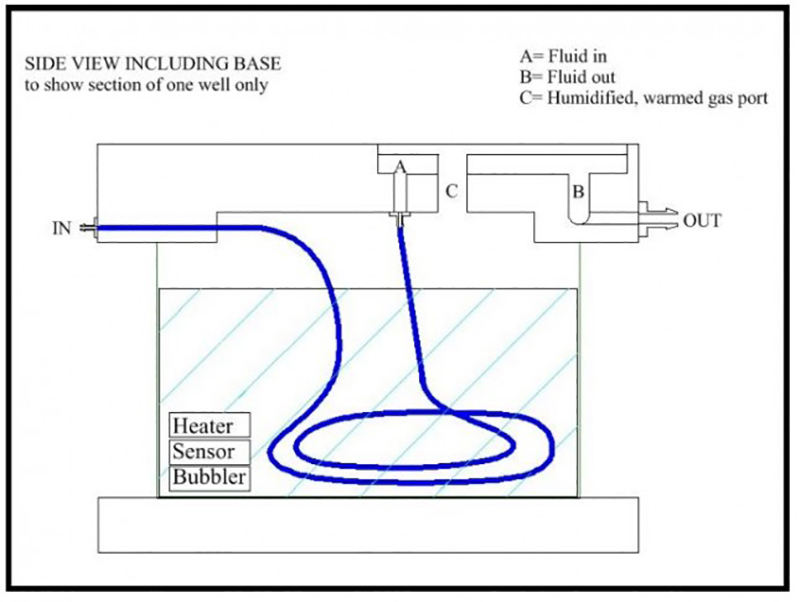 [/su_lightbox]
[/su_lightbox]
With the introduction of the BSC2-2, the original BSC2 interface chamber has been re-designed as a single channel chamber with only one perfusion line and is no longer supplied with tandem templates.
For more information and pricing, please contact Digitimer.
BSC2-2 References:-
Milner, A.J. Cummings, D.M. Stewart M.G. and Murphy K.P.S.J. (2002) Age-dependent long-term depression at CA3-CA1 synapses in mouse hippocampal slices. J Physiol 544P
Murphy, K.P.S.J., Carter, R.J., Lione, L. A., Mangiarini, L., Mahal, A., Bates, G.P., Dunnett, S.B., and Morton. J. (2000) Abnormal Synaptic Plasticity and Impaired Spatial Cognition in Mice Transgenic for Exon 1 of the Human Huntington’s Disease Mutation. J. Neuroscience 20(13):5115-5123


